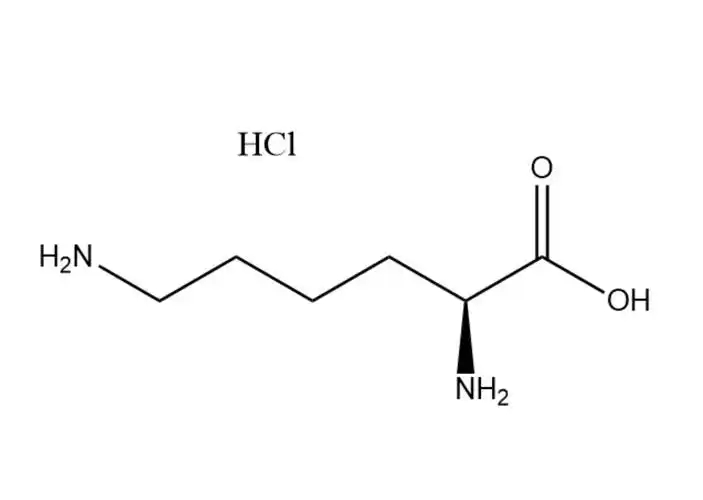
Calcium carbonate
Calcium carbonate has three different crystal structures:
Calcite: It is the most common structural form, belonging to the trigonal crystal system, and has high symmetry and stability. The crystal forms of calcite are diverse, with common ones including rhombohedrons and trigonal hedrons. In nature, marble, limestone and other materials are mainly composed of calcite.
Aragonite, also known as aragonite, belongs to the orthorhombic crystal system. Its crystal structure and physical properties are different from those of calcite. The stability of aragonite is relatively low and it can be transformed into calcite under certain conditions.
Aragonite: It is a metastable structural form and belongs to the hexagonal crystal system. Aragonite is relatively rare in nature and usually forms under specific physical and chemical conditions, such as in living organisms or during certain rapid precipitation processes.
Другие товары поставщика
|
|
Magnesium Gluconat e |
Solubility and Hydrophilicity of Magnesium GluconateWater Solubility: Highly soluble in water, with a solubility of approximately 100 g/L at 20°... |
|
|
Calcium Carbonate |
The main uses of Calcium CarbonateI. Industrial Field· Building materials: They are important raw materials for manufacturing cement, lime, ... |
|
|
Magnesium Citrate |
Magnesium influences heart rhythm, blood pressure regulation, and vascular function, making magnesium citrate relevant for cardiovascular health:Bl... |
|
|
Calcium Carbonate |
The molecular structure of Calcium CarbonateIt is formed by the combination of calcium ions (Ca²⁺) and carbonate ions (CO₃²⁻) through ion... |
|
|
Magnesium Lactate Powder |
Key Considerations for Use Safety: Generally recognized as safe (GRAS) by regulatory bodies when used within recommended limits, but excessive inta... |
Все товары поставщика
Похожие товары