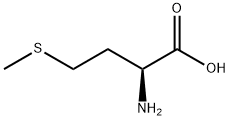
DL-methionine
The methylthio group is the most distinctive functional group in DL-methionine and can participate in substitution and addition reactions. Alkylation substitution reaction: The sulfur atom in the methylthio group has a lone pair of electrons, making it a nucleophilic site that can react with alkyl halides (e.g., methyl iodide) to form sulfonium salts. This reaction is the basis for the synthesis of methionine derivatives with enhanced biological activity. Cleavage reaction: Under the action of strong nucleophiles (e.g., sodium hydroxide in the presence of hydrogen peroxide) or enzymes (e.g., methionine lyase), the C-S bond in the methylthio group can be cleaved, producing homocysteine and methanethiol. In biological systems, this cleavage reaction is catalyzed by specific enzymes and is a key step in methionine metabolism.
Другие товары поставщика
|
|
L-Methionine in Animal Feed Additive |
L-Methionine in Animal Feed Additive Applications L-Methionine is one of the most critical nutritional additives in animal husbandry, as monogastri... |
|
|
Purchase price |
As an amphoteric compound, L-threonine can buffer pH changes in acidic or alkaline food systems: Used in beverages (e.g., sports drinks, fruit juic... |
|
|
L-Methionine |
As one of the nine essential amino acids, L-Methionine is a core building block for protein synthesis, serving two critical purposes: It acts as th... |
|
|
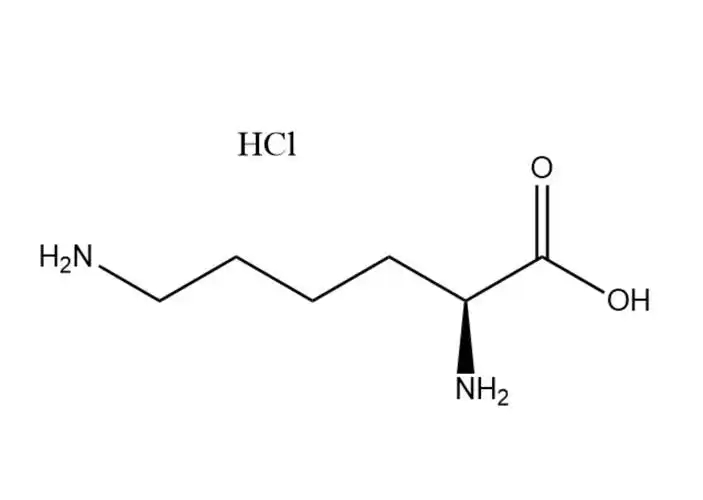
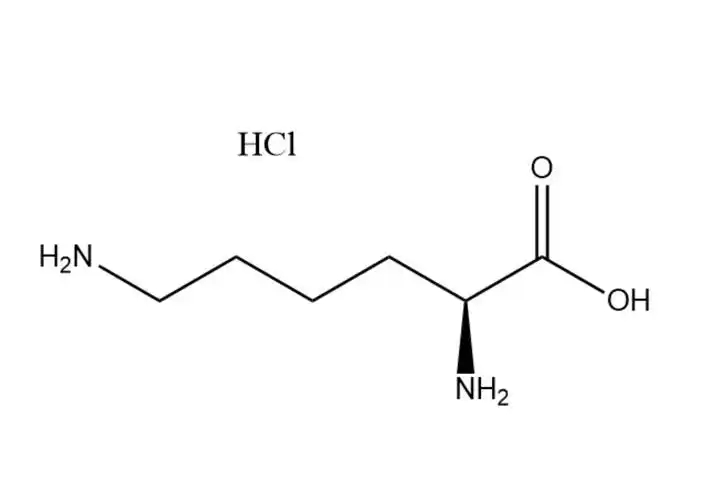
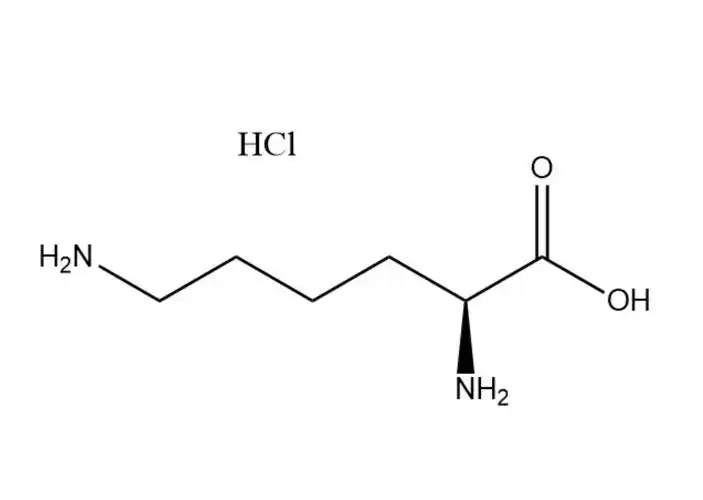
L-lysine monohydrochloride |
The free Cl⁻ ion in the hydrochloride salt form undergoes typical halide ion reactions, which are used for qualitative identification of L-lysine m... |
|
|
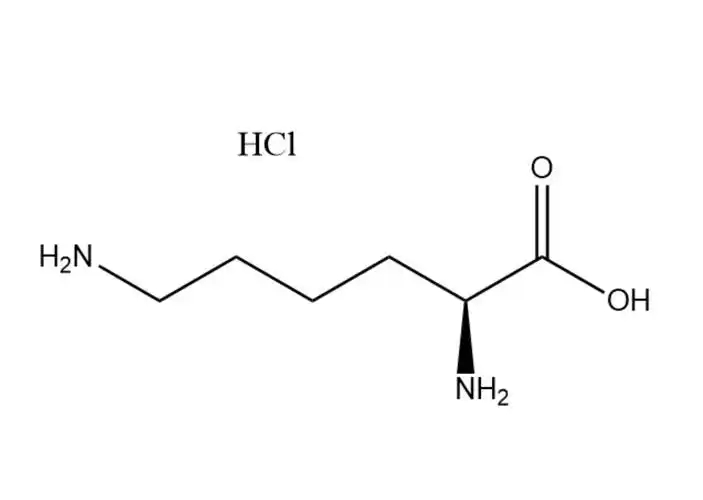
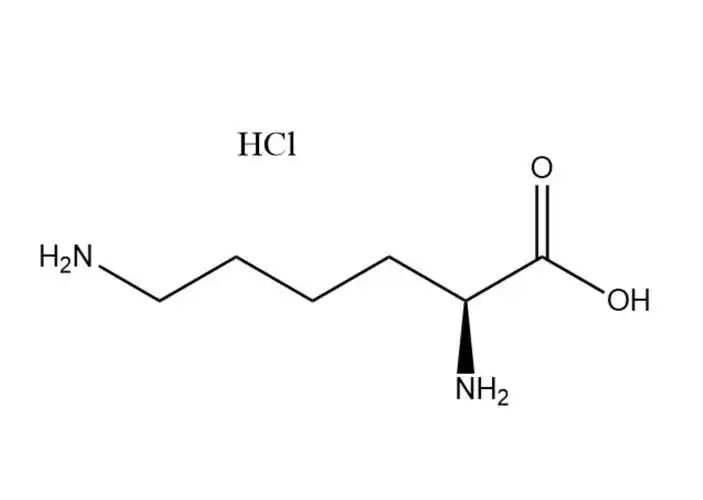
L-lysine monohydrochloride |
L-lysine monohydrochloride’s unique physical and chemical properties make it the preferred form of L-lysine for industrial and biological app... |
Все товары поставщика
Похожие товары