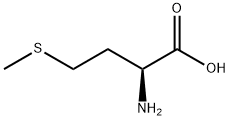
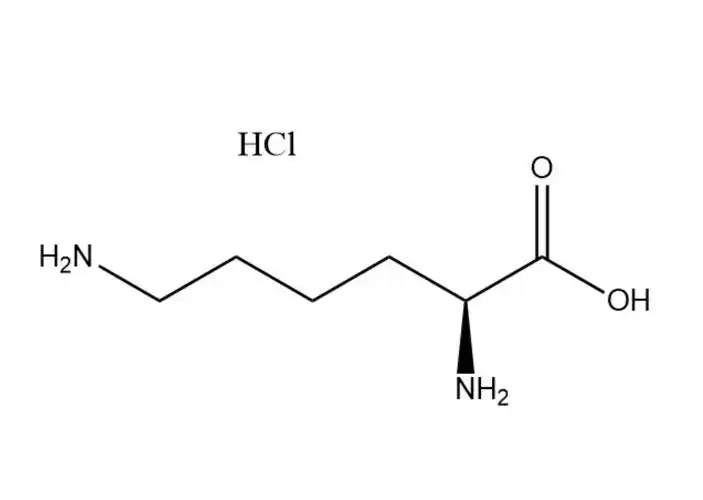
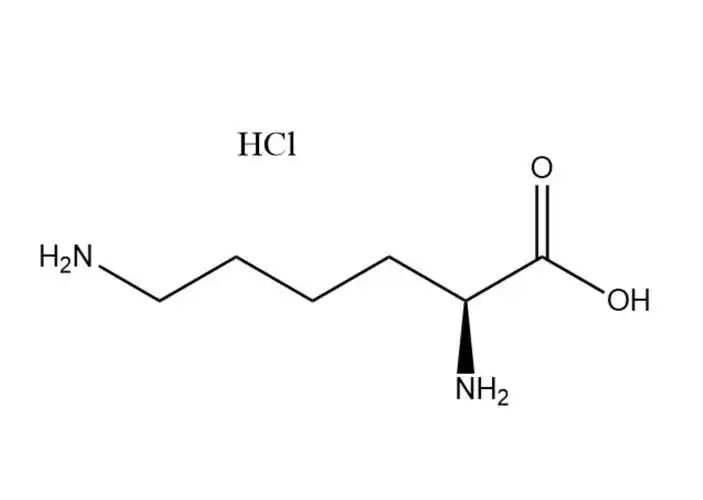
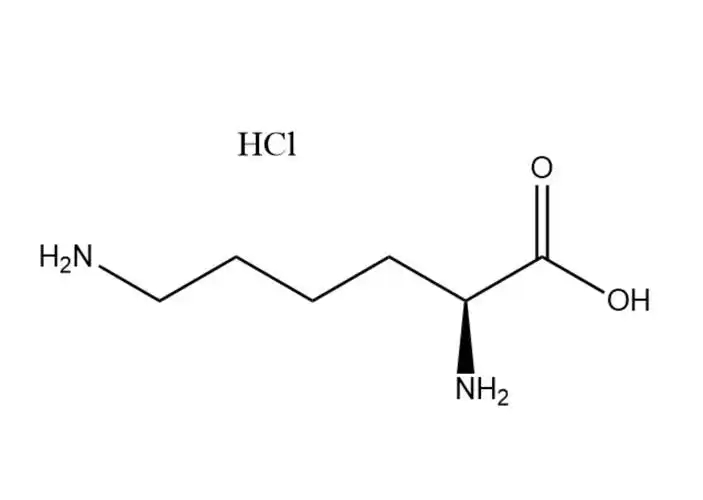
DL-methionine Partners, long-term Cooperation
The methylthio group is the most distinctive functional group in DL-methionine and can participate in substitution and addition reactions. Alkylation substitution reaction: The sulfur atom in the methylthio group has a lone pair of electrons, making it a nucleophilic site that can react with alkyl halides (e.g., methyl iodide) to form sulfonium salts. This reaction is the basis for the synthesis of methionine derivatives with enhanced biological activity. Cleavage reaction: Under the action of strong nucleophiles (e.g., sodium hydroxide in the presence of hydrogen peroxide) or enzymes (e.g., methionine lyase), the C-S bond in the methylthio group can be cleaved, producing homocysteine and methanethiol. In biological systems, this cleavage reaction is catalyzed by specific enzymes and is a key step in methionine metabolism.
Other supplier products
|
|
Bulk supply L-Threonine |
Chemical Properties of L-Threonine Amino Acid Structure Contains an amino group (-NH₂), a carboxyl group (-COOH), and a hydroxyl-containing side ch... |
|
|
High quality L-proline |
Metabolism and Safety of L-prolineMetabolic pathway: Mainly metabolized in the liver and kidneys, it is converted into glutamate hemialdehyde throu... |
|
|
Sucralose 56038-13-2 |
Sucralose is a white powder product that readily dissolves in water, ethanol, and methanol. It is currently the only functional sweetener produced ... |
|
|
L-Methionine 63-68-3 |
Core Biological Functions of L-Methionine Protein Synthesis Precursor: As an essential amino acid, L-Methionine is a fundamental building block for... |
|
|
High quality L-tryptophan |
Beyond natural occurrence, L-tryptophan is sometimes added to fortified foods and functional products to enhance nutritional value or provide speci... |
All supplier products
Same products