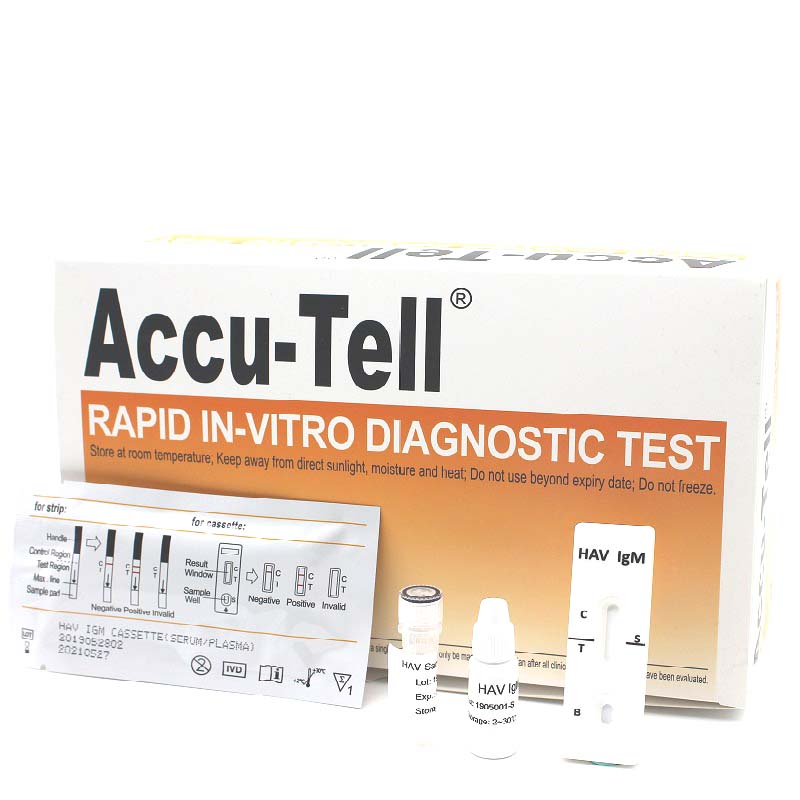
Accu-Tell® HAV IgM Rapid Test Cassette (Serum/Plasma)
Accu-Tell®HAV IgM Rapid Test Cassette (Serum/Plasma) is a rapid chromatographic immunoassay for the qualitative detection of IgM antibody to Hepatitis A virus (HAV) in serum or plasma.
CATALOG
|
Product Name |
Specimen |
Catalog No. |
Certificate |
Quantity per box |
|
HAV IgM Rapid Test Cassette |
Serum/Plasma |
ABT-IDT-B218 |
CE |
40T/20T |
KEY POINTS
Detection for:IgM anti-HAV in serum or plasma;
Specimen volume:50 μl;
Reading time:20 minutes;
Relative Sensitivity:>99.9% (95%CI*: 88.7%-100%);
Relative Specificity:99.1% (95%CI*: 96.8%-99.9%);
Accuracy:99.2% (95%CI*: 97.1%-99.9%).
INTRODUCTIONof
HAV is a positive RNA virus, a unique member of picornavirdae. Its transmission depends primarily on serial transmission from person to person by the fecal-oral route. Although hepatitis A is not ordinarily a sexually transmitted disease, the infection rate is high among male homosexuals, as result of oral-anal contact.
The presence of specific anti-HAV IgM in blood samples suggests acute or recent HAV infection. The IgM antibody rapidly increases in titer over a period of 4-6 weeks post infection, and then declines to non-detectable levels within 3 to 6 months in most patients.
INTENDED USE
ACCU-TELL®(Serum/Plasma) is a rapid chromatographic immunoassay for the qualitative detection of IgM antibody to Hepatitis A virus (HAV) in serum or plasma.
PERFORMANCE CHARACTERISTICSof
Sensitivity and Specificity
ACCU-TELL®HAV IgM Rapid Test Cassette (Serum/Plasma) was compared with a leading commercial ELISA HAV IgM test; the results show that ACCU-TELL® HAV IgM Cassette (Serum/Plasma) has a high sensitivity and specificity.
|
Method |
EIA |
Total Results |
||
|
ACCU-TELL®HAV IgM Cassette (Serum/Plasma) |
Results |
Positive |
Negative |
|
|
Positive |
25 |
2 |
27 |
|
|
Negative |
0 |
221 |
221 |
|
|
Total Results |
25 |
223 |
248 |
|
Relative Sensitivity: >99.9% (95%CI*: 88.7%-100%)
Relative Specificity: 99.1% (95%CI*: 96.8%-99.9%)
Accuracy: 99.2% (95%CI*: 97.1%-99.9%)
*95% Confidence Intervals
LIMITATIONS
- We are also
Note: The above information is for reference use only. Please refer to the product insert provided with the products before use.
Send product request
Other supplier products
| Accu-Tell® HAV IgM Rapid Test Cassette (Serum/Plasma) | Accu-Tell®HAV IgM Rapid Test Cassette (Serum/Plasma) is a rapid chromatographic immunoassay for the qualitative detection of IgM antibody to H... | |
| 17.Accu-Tell® HBcAb Rapid Test Cassette (Serum/Plasma) | Accu-Tell®HBcAb Rapid Test Cassette (Serum/Plasma) is a rapid chromatographic immunoassay for the qualitative detection of Hepatitis B Core A... | |
| Accu-Tell® Multi-Drug Saliva Rapid Test Cassette | Accu-Tell®for AMP/MET/COC/OPI/THC/PCP/MTD/MDMA/OXY/COT/BZO/KET/6-MAM is a lateral flow chromatographic immunoassay for the qualitative dete... | |
| Accu-Tell® Multi-line Drug Rapid Test Cassette (Urine) | Accu-Tell®Multi-line Drug Cassette (Urine) is a rapid chromatographic immunoassay for the qualitative detection of multiple drugs and drug ... | |
| Accu-Tell® Multi-Drug Rapid Test Panel (Urine) | Accu-Tell®Multi-Drug Rapid Test Panel (Urine) is a rapid chromatographic immunoassay for the qualitative detection of multiple drugs and dr... |
Same products
| Digital Construction | Seller: FJ Dynamics Co., Ltd. | We don’t do construction, but we are transforming construction. FJD machine control system ... | |
| FJD WiFi Camera for Autosteering Kit | Seller: FJ Dynamics Co., Ltd. | $299.00 Tax excluded. Taxes and Shipping calculated at checkout Quantity: 1 Compatible wi... | |
| Extension Sets | Seller: Shanghai Kohope Medical Devices Co., Ltd. | Now, we provide various types of extension sets, including iv extension set with injection port, ... | |
| Next Generation Sequencing Platform | Seller: CapitalBio Technology Co, Ltd. | NGSPlatforms Overview A high-throughput sequencingplatform is built around the Bioelectron 4000 ... | |
| Chicken IgY | Seller: Hanzghou Frenovo Biotech Co.,Ltd. | The IgY isotype control could be used in FC, ELISA and Flisa experiments. Each batch of the produ... |