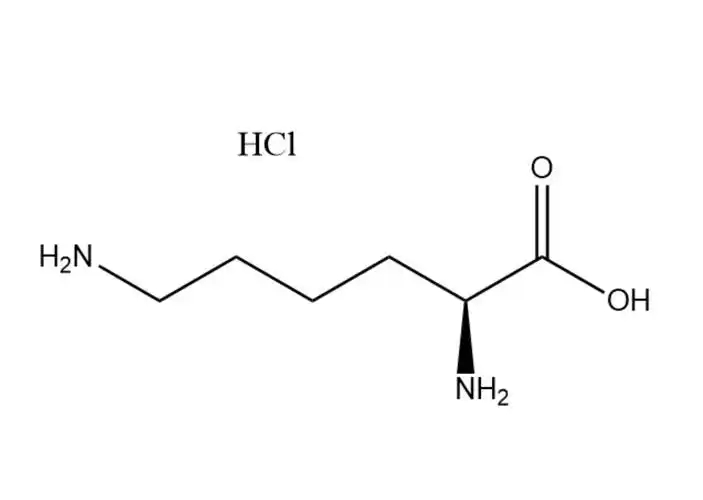
Calcium carbonate


Calcium carbonate can be classified into light calcium carbonate, heavy calcium carbonate and active calcium carbonate based on different production methods.
The preparation method of light calcium carbonate (referred to as light calcium) is to calcine raw materials such as limestone to generate lime (mainly composed of calcium oxide) and carbon dioxide, then add water to digest the lime to form lime milk (mainly composed of calcium hydroxide), and then introduce carbon dioxide to carbonize the lime milk to generate calcium carbonate precipitate. Finally, it is obtained through dehydration, drying and grinding. It can be obtained by first conducting a double decomposition reaction between sodium carbonate and calcium chloride to form calcium carbonate precipitate, and then through dehydration, drying and grinding. It is mainly used as a filler in industries such as rubber, plastic, papermaking, coatings and inks, and can also be used in daily chemical products such as tooth powder, toothpaste and cosmetics. It can also be used in organic synthesis, metallurgy, glass and asbestos production.
The preparation method of heavy calcium carbonate (referred to as heavy calcium) is to directly crush natural calcite, limestone, chalk, shells, etc. by mechanical means. It is an excellent filler and performance modifier, and is widely used in plastics, rubber, papermaking, coatings, feed, medicine, daily chemical industry, glass, ceramics and other fields.
The preparation method of active calcium carbonate (referred to as active calcium) is to modify the surface of light calcium or heavy calcium with surface modifiers to obtain it. After the modification treatment, the surface of the calcium carbonate powder forms a special coating structure, which can significantly improve the dispersion and affinity in polymer matrices such as polyolefins, and can also produce interface interaction with the polymer matrix. Thereby enhancing the impact resistance strength of the products, it is an excellent incremental filling additive.
Другие товары поставщика
|
|
Manganese Gluconate |
Product Name:Manganese Gluconate Description Molecular formula:C12H22MnO14 • 2H2O Molecular weight:481.27 Manganese assay:11.43% Shelf life:24... |
|
|
Magnesium Gluconat e |
Solubility and Hydrophilicity of Magnesium GluconateWater Solubility: Highly soluble in water, with a solubility of approximately 100 g/L at 20°... |
|
|
Market quotation |
Stability and Compatibility of Magnesium Gluconate Chemical Stability: Stable under normal storage conditions when kept dry, but may degrade in the... |
|
|
Magnesium Citrate |
Magnesium citrate is particularly valuable in foods designed for specific dietary needs: Low-Sodium Products: As a salt substitute or complement, i... |
|
|
Monosodium Fumarate |
Product Name:Monosodium Fumarate Description Molecular formula:C4H3NaO4 Molecular weight:138.05 Monosodium Fumarate assay:98.0~102.0% Shelf life:24... |
Все товары поставщика
Похожие товары