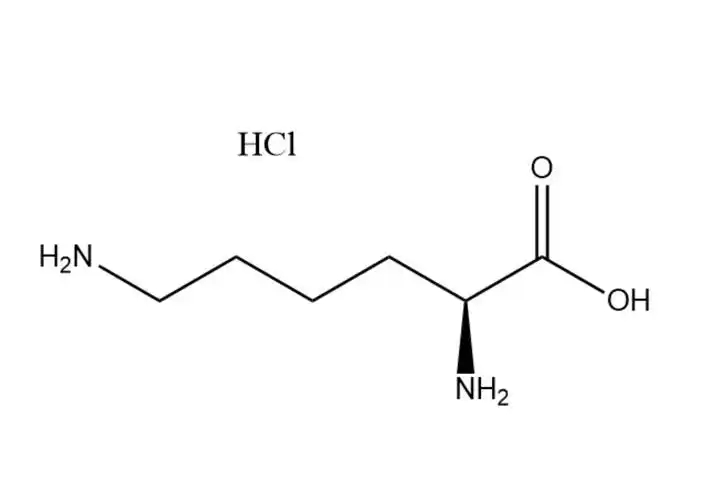
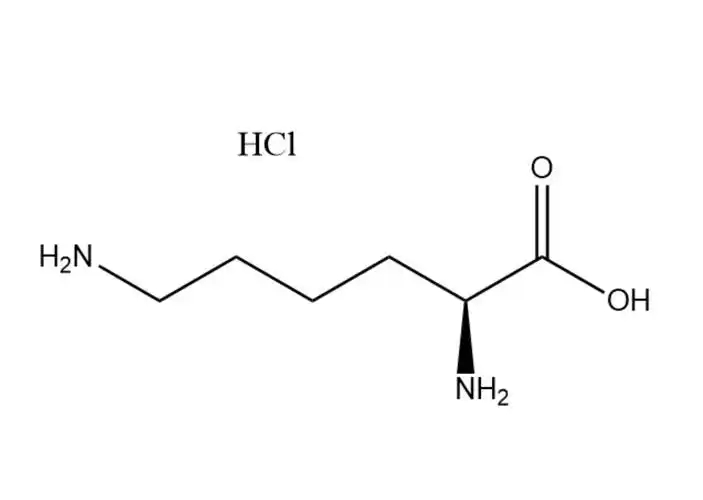
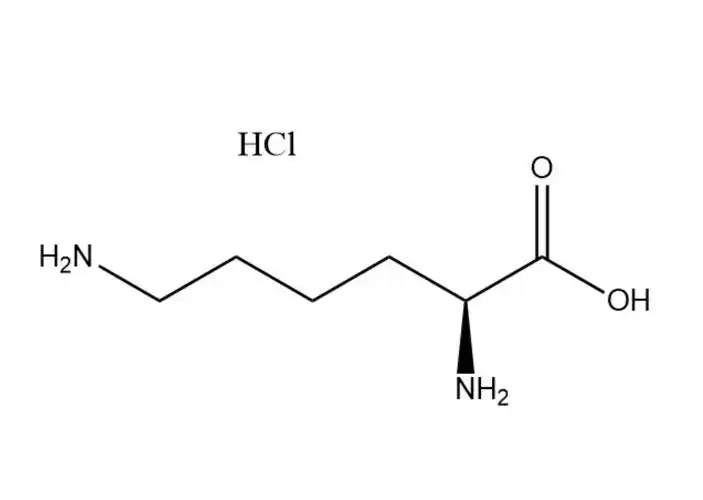
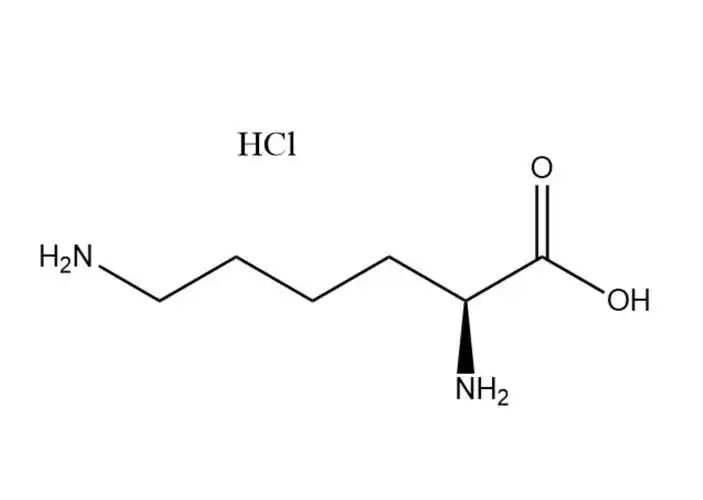
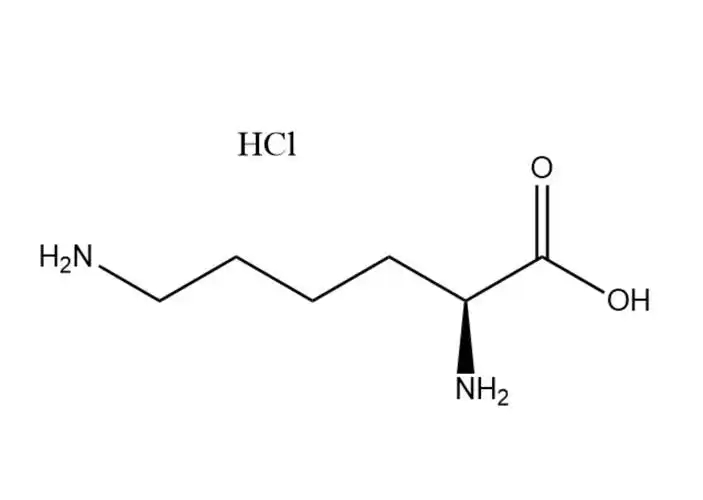
L-Lysine monohydrochloride
L-Lysine monohydrochloridehas a higher aqueous solubility than free L-lysine: ~70 g/100 mL at 25 °C (vs. ~63 g/100 mL for free L-lysine), with solubility increasing significantly with temperature (up to 110 g/100 mL at 60 °C). Its dissolution is rapid, endothermic, and free of precipitation in dilute to high-concentration aqueous solutions (up to 60% w/v). This advantage makes it ideal for:
- Liquid formulations (e.g., oral nutritional supplements, animal drinking water additives, pharmaceutical syrups) where high solubility and clear solutions are required.
- Industrial production processes (e.g., fermentation downstream purification, formulation blending) that demand fast dissolution and uniform dispersion in aqueous systems.
In contrast, free L-lysine has lower solubility and may form supersaturated solutions that precipitate during storage, leading to formulation instability.
Другие товары поставщика
|
|
DL-Alanine |
DL-Alanine is recognized asGenerally Recognized as Safe (GRAS)by major regulatory bodies (e.g., FDA in the U.S., EFSA in the EU, and CFSA in China)... |
|
|
Glycine |
Glycine has three reaction centers: amino group, carboxyl group, and methylene group. It has typical and diverse chemical properties.1. Ampholytic ... |
|
|
Glycine |
Glycine is a very simple α-amino acid with the widest range of applications among the non-essential amino acids in the human body. Due to its... |
|
|
DL-Methionine |
Hygroscopicity and Bulk Properties of DL-MethionineHygroscopicity: DL-methionine has weak hygroscopicity. Under ambient conditions (25 °C, rela... |
|
|
DL-Methionine |
Redox Properties of DL-MethionineThemethylthio group (-SCH3)in the side chain of DL-methionine is the core moiety responsible for its redox reactiv... |
Все товары поставщика
Похожие товары