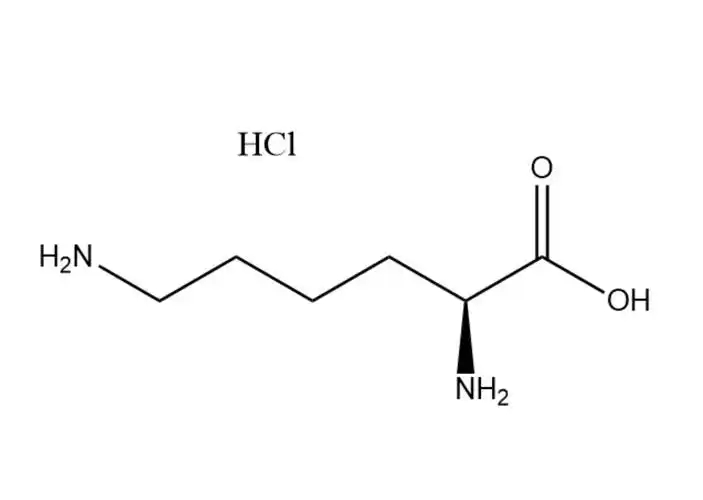
Magnesium Citrate
Chemical Formula: C₄H₃NaO₄
It is formed by the partial neutralization of fumaric acid (HOOC-CH=CH-COOH), where one carboxyl group (-COOH) loses a hydrogen ion (H⁺) and binds to a sodium ion (Na⁺), resulting in the structure: NaOOC-CH=CH-COOH.
Molecular Weight: Approximately 138.06 g/mol (anhydrous form).
Stability: Relatively stable under normal conditions (room temperature, dry storage). It may decompose at high temperatures (above 200°C) or in strongly alkaline environments, releasing carbon dioxide or other byproducts.
Reactivity: As an acid salt, it can react with bases to form disodium fumarate (NaOOC-CH=CH-COONa) and water. It also interacts with carbonates (e.g., baking soda) to release carbon dioxide, a property used in leavening baked goods.
Other supplier products
|
|
Calcium carbonate |
Calcium carbonate has three different crystal structures:Calcite: It is the most common structural form, belonging to the trigonal crystal system, ... |
|
|
Supermicro Zinc Citrate |
Product Name:Supermicro Zinc Citrate Description Raw material :Zinc Citrate Particle size range:120 - 1100 mesh Shelf life:24 months: Packing :25kg... |
|
|
Magnesium Citrate |
Magnesium citrate is particularly valuable in foods designed for specific dietary needs: Low-Sodium Products: As a salt substitute or complement, i... |
|
|
Calcium carbonate |
Calcium carbonate can be classified into light calcium carbonate, heavy calcium carbonate and active calcium carbonate based on different productio... |
|
|
Sodium Malate |
Product Name:Sodium Malate Description Molecular formula:C4H4Na2O5 Molecular weight:178.05 Sodium Malate assay:98.0~102.0% Shelf life:24 month CAS ... |
供应产品
Same products