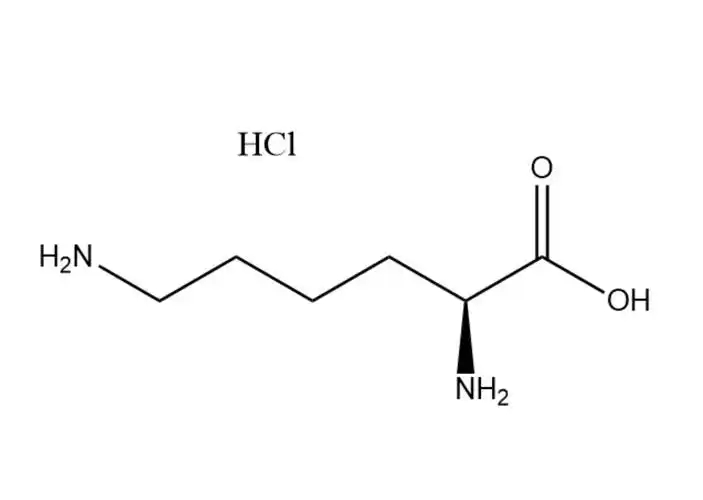
Magnesium Citrate Raw Material
Monosodium Fumarateis a sodium salt derived from fumaric acid, with distinct chemical and physical properties that underpin its practical applications. Below are its key basic properties:
- Chemical Formula: C₄H₃NaO₄
It is formed by the partial neutralization of fumaric acid (HOOC-CH=CH-COOH), where one carboxyl group (-COOH) loses a hydrogen ion (H⁺) and binds to a sodium ion (Na⁺), resulting in the structure: NaOOC-CH=CH-COOH.
- Molecular Weight: Approximately 138.06 g/mol (anhydrous form).
- Stability: Relatively stable under normal conditions (room temperature, dry storage). It may decompose at high temperatures (above 200°C) or in strongly alkaline environments, releasing carbon dioxide or other byproducts.
- Reactivity: As an acid salt, it can react with bases to form disodium fumarate (NaOOC-CH=CH-COONa) and water. It also interacts with carbonates (e.g., baking soda) to release carbon dioxide, a property used in leavening baked goods.
- Appearance: Typically a white, odorless crystalline powder or fine granules.
- Solubility: Highly soluble in water (approximately 100 g/L at 20°C), which makes it easy to dissolve in liquid formulations (e.g., beverages, syrups). It is less soluble in organic solvents like ethanol.
- Taste: Has a slightly tart, acidic flavor (milder than fumaric acid itself), contributing to its use as a flavor enhancer in foods.
- pH: Aqueous solutions are weakly acidic, with a pH of around 3–4 (depending on concentration), due to the remaining carboxyl group (-COOH) that can release H⁺ ions.
It has low hygroscopicity, meaning it does not readily absorb moisture from the air. This makes it stable in dry storage and suitable for use in powdered or granular food products without clumping.
These properties—solubility, mild acidity, stability, and low hygroscopicity—make monosodium fumarate particularly useful as a food additive, pH adjuster, and minor industrial ingredient.
Other supplier products
|
|
Magnesium Citrate |
Chemical Formula: C₄H₃NaO₄
It is formed by the partial neutralization of fumaric acid (HOOC-CH=CH-COOH), where one carboxyl group (-COOH) loses a ... |
|
|
Calcium carbonate |
Calcium carbonate, also known as limestone or stone powder, is a common chemical substance on Earth. It belongs to inorganic salt minerals, is alka... |
|
|
Magnesium Citrate |
Magnesium is involved in neurotransmitter synthesis and regulation, supporting brain function and mood: Stress and anxiety management: Magnesium mo... |
|
|
Calcium Carbonate |
The molecular structure of Calcium CarbonateIt is formed by the combination of calcium ions (Ca²⁺) and carbonate ions (CO₃²⁻) through ion... |
|
|
Copper Gluconate |
Product Name:Copper Gluconate Description Molecular formula:C12H22CuO14 Molecular weight:453.84 Copper assay:14 % Shelf life: 24 month CAS NO.:527-... |
供应产品
Same products