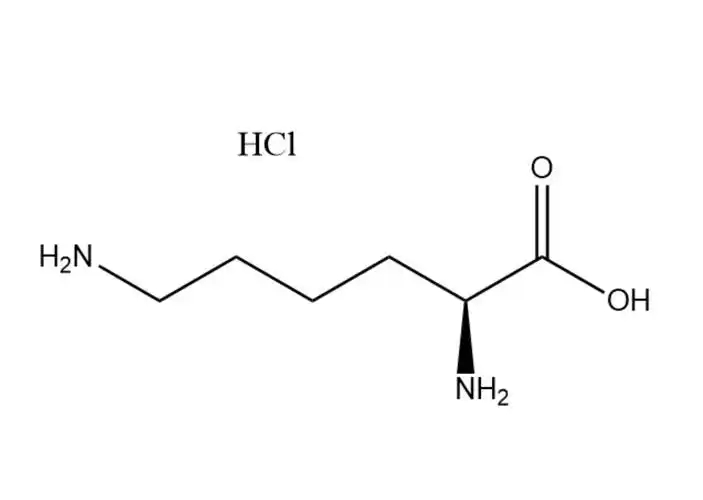
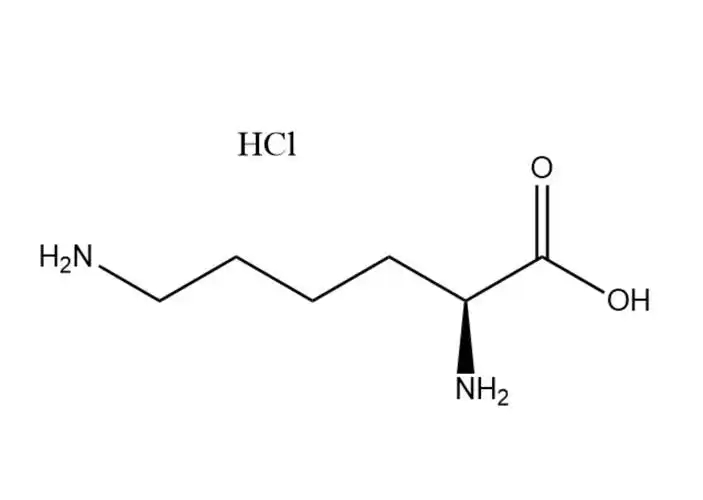
L-lysine monohydrochloride
The free Cl⁻ ion in the hydrochloride salt form undergoes typical halide ion reactions, which are used for qualitative identification of L-lysine monohydrochloride (per USP/EP/ChP standards): Reacts with silver nitrate (AgNO3) in aqueous solution to form a white, insoluble precipitate of silver chloride (AgCl), which is insoluble in dilute nitric acid— a classic test for Cl⁻ ions. No redox reaction of Cl⁻ under normal conditions (Cl⁻ is stable and non-toxic).
Other supplier products
|
|
DL-Alanine |
DL-Alanine’s uses span multiple fields, leveraging its amino acid properties and cost-effectiveness (it is often cheaper to produce than pure... |
|
|
L-proline |
L-proline, to ensure its stable quality and performance, certain conditions need to be met during storage. The following is a detailed introduction... |
|
|
L-proline |
The main properties of L-prolineSolubilityIt is readily soluble in water (about 162 g/L, 25°C), slightly soluble in ethanol, and insoluble in e... |
|
|
DL-methionine |
The α-amino group in DL-methionine has the typical reactivity of a primary amine and can participate in acylation, alkylation, and condensati... |
|
|
DL-Alanine |
DL-Alanine can interact with proteins and starches in food, modifying their structure to improve texture and stability—especially in processe... |
供应产品
Same products