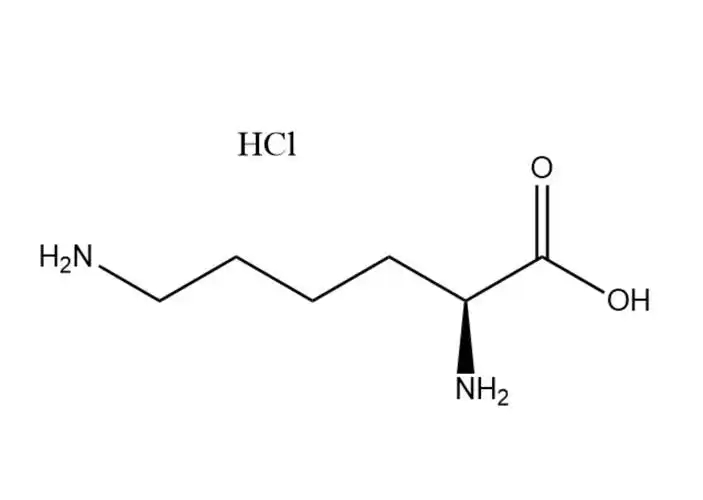
Calcium Carbonate
The molecular structure of Calcium Carbonate
It is formed by the combination of calcium ions (Ca²⁺) and carbonate ions (CO₃²⁻) through ionic bonds and belongs to ionic crystals.
It exists in nature in various crystal forms. Common ones include:
Calcite: Trigonal crystal system, with a compact crystal structure and a relatively high density (2.71g /cm³), is the most stable crystal form of calcium carbonate.
Aragonite: Orhombic crystal system, with a slightly higher density (2.93 g/cm³). Thermodynamically, it is less stable than calcite and can transform into calcite over a long period of time.
Vaterite: Hexagonal crystal system, rare, metastable crystal form, easily transforms into calcite or aragonite.
Другие товары поставщика
|
|
Compound nutritional fortifier (Biotin dilution) |
Product Name:Compound nutritional fortifier (Biotin dilution) Description Raw material :D-biotin, calcium hydrogen phosphate, maltodextrin Assay:1%... |
|
|
Magnesium Citrate |
Magnesium citrate influences the texture and shelf stability of food products through its interaction with other ingredients:Processed Meats and Se... |
|
|
Magnesium Gluconate |
Hygroscopicity and Moisture Absorption of Magnesium Gluconate Hygroscopic Nature: Magnesium gluconate is moderately hygroscopic, meaning it absorbs... |
|
|
Magnesium Citrate |
Magnesium citrate is widely recognized for its role in supporting digestive regularity:Osmotic laxative: When taken in higher doses, magnesium citr... |
|
|
Calcium Carbonate |
Safety and precautions of Calcium CarbonateDust hazard: Long-term inhalation of calcium carbonate dust may cause respiratory irritation (such as pn... |
Все товары поставщика
Похожие товары