DL-Alanine
DL-Alanine is not naturally abundant in living organisms (most life forms primarily use L-Alanine as a building block for proteins). It is mainly producedsyntheticallythrough two common methods:
- Chemical Synthesis: Typically via the "Strecker synthesis" (reacting acetaldehyde with ammonia and cyanide to form an amino nitrile, which is then hydrolyzed to DL-Alanine) or the "amination of pyruvate" (reacting pyruvic acid with ammonia under catalytic conditions to generate a racemic mixture of alanine).
- Biotechnological Production: Some microbial fermentation processes (using engineered bacteria) can produce DL-Alanine by converting glucose or other carbon sources, though this method is less common than chemical synthesis.
Другие товары поставщика
|
|
L-Cysteine hydrochloride monohydrate |
Packaging Requirements1. Primary PackagingSealable Bags/Bottles: Use hermetically sealed bags (e.g., foil-lined packages) or airtight bottles with ... |
|
|
L-threonine |
L-threonine can enhance the flavor and seasoning Umami and Sweetness Modulation: L-Threonine has a slightly sweet taste and can enhance the overall... |
|
|
Sucralose |
Sucralose is widely used in processed foods such as beverages, chewing gum, dairy products, dried fruits, jelly puddings, bread, pastries, cookies,... |
|
|
DL-Alanine |
The defining feature of a direct food preservative is its ability to target and inhibit the metabolic processes or structural integrity of microorg... |
|
|
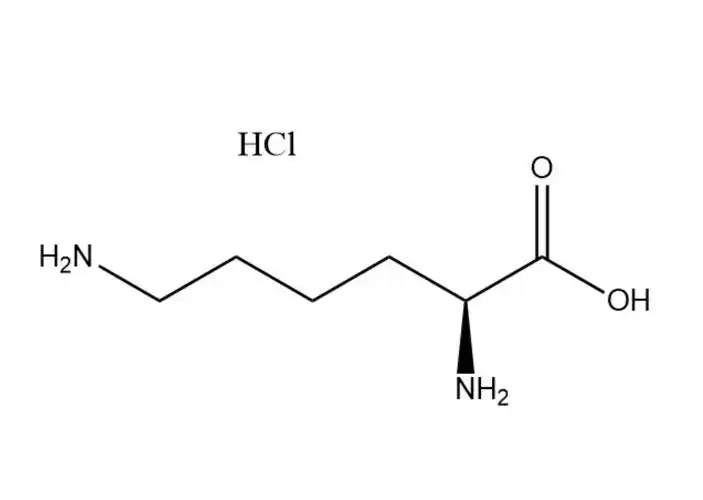
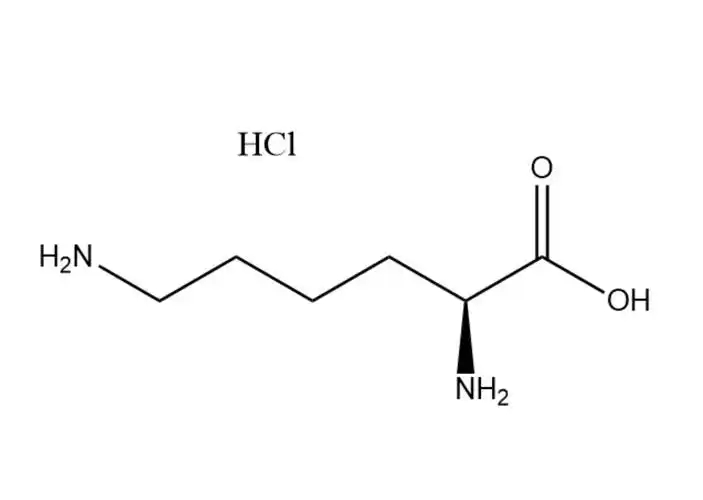
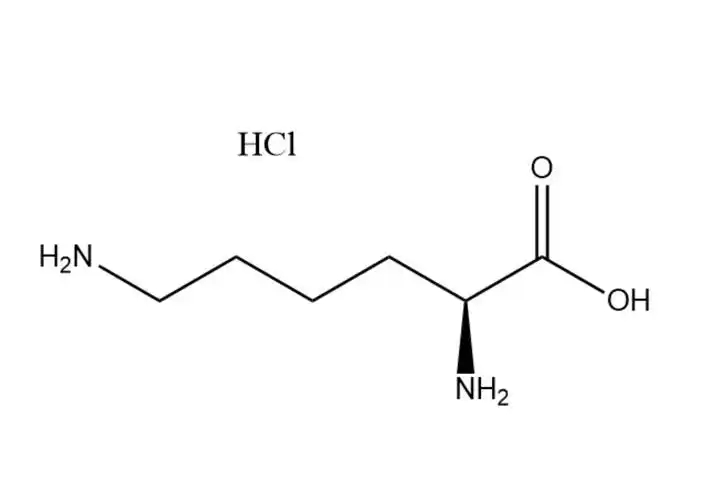
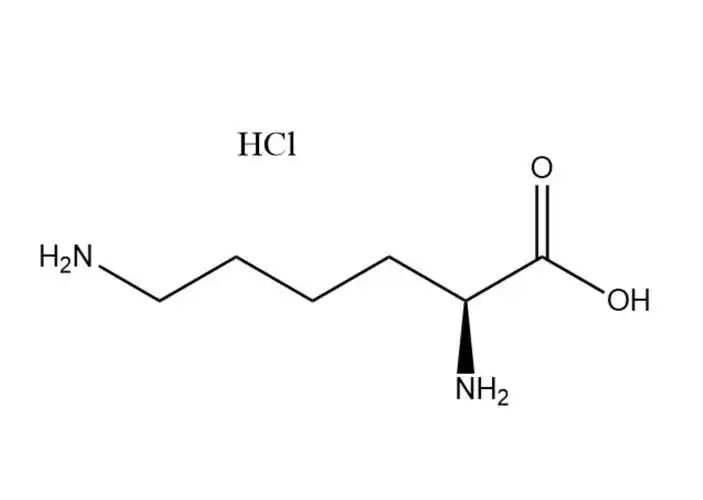
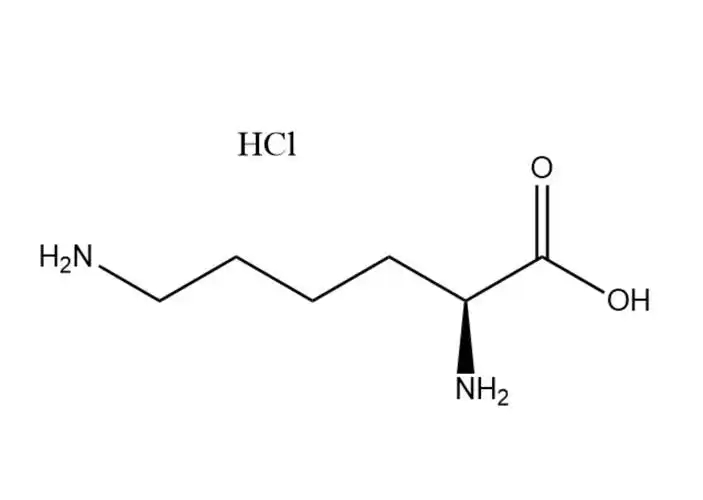
L-Lysine monohydrochloride |
The most prominent performance advantage ofL-Lysine monohydrochloride is its optimized physical and chemical properties that eliminate the critical... |
Все товары поставщика
Похожие товары